F
c-Engineered Antibody Expression Vectors with enhanced Fc-effector function are now available in store. These include human IgG1 and IgG4 heavy chains with increased or decreased ADCC and CDC, an aglycosylated human IgG1 heavy chain variant, and a human IgG1 with extended serum half-life.
New Products
Fc-Engineered Antibody Expression Vectors
New Phagemid Vectors
P
hagemid vectors pADL-43 and pADL-23Chlo are now available in store. The pADL-23Chlo™ phagemid is a variant of pADL-23c™ that uses a different antibiotic marker. pADL™-43 allows display on a truncated version of pIII, similarly to pComb3 vector. Both vectors completes the series of phagemids offered by Antibody Design Labs.
MIM™ Peptide Phage Libraries
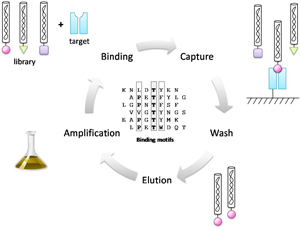
The MIM™ Phage Display Peptide Libraries are full-phage libraries consisting of ~109 random peptides displayed as an N-terminal fusion protein with the minor coat protein pIII. These libraries are similar to the libraries originally developed by George Smith and are currently available as a 12-mer (NH2-X12GGGSGPGGLRGGS-pIII, MIM-12) and cyclic 10-mer (NH2-AEGCX10C GGGSGPGGLRGGS-pIII, MIM-C10) phage libraries. A lysine conveniently located in the linker allows for the more efficient trypsin elution.
Novel Mammalian Expression Vector Series
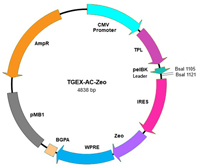
The TGEX™ Vector Series is getting an upgrade! The new series has been optimized for improved expression of recombinant antibodies and now offers the selection of stable pools and stable transformants using antibiotics. The series offers a complete coverage of human, mouse and rabbit IgG isotypes as well as several vectors for the expression of Fab fragments and Fc fusions.
fADL-2blue: A Trypsin-Sensitive Helper Phage Vector
hage vector fADL-2blue is now available in store. The fADL-2blue™ phage vector continues the fADL phage vector series by bringing trypsin-sensitivity for elution together with ampicillin-resistance and blue colonies that together limit contaminations by other phage. fADL-2blue also has an improved virion production and a higher content of DNA in its replicative form. fADL-2blue™ rivals with the best vectors build by George Smith, the inventor of phage display.
CM13d3 pIII-Defective Helper Phage
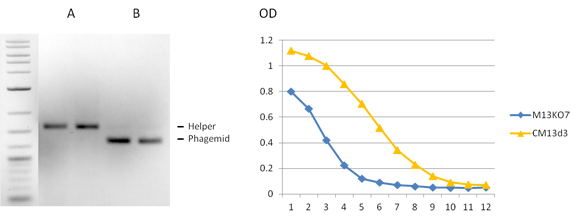
CM13d3 is a novel helper phage engineered for multivalent phage display. CM13d3 lacks a functional gene III driving the display of pIII-fusion proteins expressed by the phagemid. CM13d3 is similar to pIII-defective helpers such as M13KO7d3, Hyperphage, Ex-Phage or Phaberge that have been shown to increase display of scFv by a factor up to 100-fold and more. CM13d3 is highly infective, making it de facto the best choice for building libraries.
Fluorescent GFP Reporter for Transfection Efficiency

The TGEX™-eGFP vector is a green fluorescent reporter designed to monitor transfection efficiency during transient gene expression in mammalian cell suspensions culture. TGEX™-eGFP vector can be used as a single component during transfection to analyze transfection efficiency or at a lower level, between 5% and 10% of the total DNA, to measure efficiency during transient gene expression.
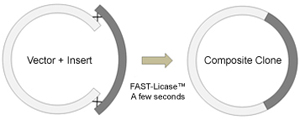
New: FAST-Licase Cloning
ntibody Design Labs is proud to announce the availability of the FAST-Licase cloning system. FAST-licase enables the ultra-fast cloning in just a few seconds of two or more DNA fragments by homologous recombination. This novel toolbox for the molecular biologist rethink traditional cloning with immense savings of both time and money.
New Phagemid Vector pADL-100 Series for Peptide Display
hage vector fADL-2blue is now available in store. The fADL-2blue™ phage vector continues the fADL phage vector series by bringing trypsin-sensitivity for elution together with ampicillin-resistance and blue colonies that together limit contaminations by other phage. fADL-2blue also has an improved virion production and a higher content of DNA in its replicative form. fADL-2blue™ rivals with the best vectors build by George Smith, the inventor of phage display.
Validating Phage Clones as scFv-Fc Fusions
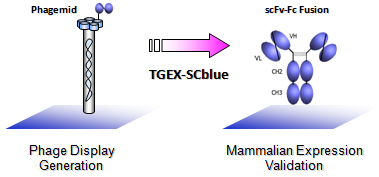
The TGEX-SCblue vector let you transfer scFv fragments seamlessly between phage clones and an Fc fusion cassette thanks to a common double SfiI site, enabling easy and rapid validation of new antibodies as scFv-Fc fusions.
