Llama Single Domain Antibody Platform
Antibody Design Labs offers a complete solution for generating llama antibodies.
Applications of Single Domain Antibodies
What are Single Domain Antibodies?
S ingle domain antibodies, also known as VHH, are made of one single antibody binding domain without associated light chain. This configuration, almost unique in the world of antibodies, confers a number of highly desirable properties. From a biotechnology point of view, sdAb are easier to clone, easier to engineer, and easier to incorporate or fuse with other antibody domains for example to create bispecific or trispecific antibodies; they are highly stable and can refold spontaneously following denaturation; they do express extremely well in many systems, including bacterial hosts and mammalian cells. Their small size makes them attractive from a therapeutic standpoint; they do penetrate tissues and cross natural barriers more easily than classical antibodies and can reach hidden epitopes located deeper in protein structure, for example in deep grooves located on the virus surface or the catalytic site of enzymes.
ingle domain antibodies, also known as VHH, are made of one single antibody binding domain without associated light chain. This configuration, almost unique in the world of antibodies, confers a number of highly desirable properties. From a biotechnology point of view, sdAb are easier to clone, easier to engineer, and easier to incorporate or fuse with other antibody domains for example to create bispecific or trispecific antibodies; they are highly stable and can refold spontaneously following denaturation; they do express extremely well in many systems, including bacterial hosts and mammalian cells. Their small size makes them attractive from a therapeutic standpoint; they do penetrate tissues and cross natural barriers more easily than classical antibodies and can reach hidden epitopes located deeper in protein structure, for example in deep grooves located on the virus surface or the catalytic site of enzymes.
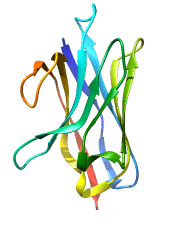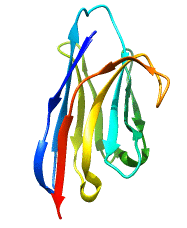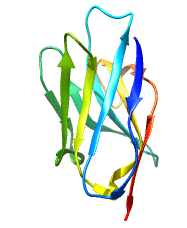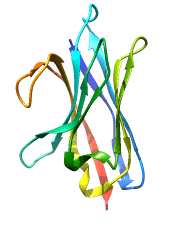 Structure of Single Domain Antibodies
Structure of Single Domain Antibodies
Single domain antibodies are monovalent and antigen-specific. Like all immunoglobulin domains, the variable domain of sdAb consists of a pair of β-sheets held together by a disulfide bridge. The loops bridging the antiparallel β-strands of the two sheets create the complementarity-determining region or CDR of the antigen binding site. Therefore VHH have only 3 CDRs instead of 6 in classical antibodies. The absence of light chain and exposure of the normally hydrophobic domain interface are compensated by a number of structural characteristics; these include the presence of specific amino acids, in particular charged amino acids, in key positions of frame 2 and, in the case of naturally occurring VHH, a particularly extended CDR3 that masks in part the hydrophobic interface.
Advantages of Single Domain Antibodies
- High affinity Small Size Antibody. Naturally occurring smallest known form of antibody, sdAb are only 13 kD and around 120 a.a. Their minimal size still allows for high specific with subnanomolar affinity.
- Good Physicochemical Properties. sdAb have a great stability with improved resistance to pH variations and heat. They can reversibly fold after heat-induced denaturation. sdAb are good candidates for intrabody development to target intracellular proteins and show great potential for oral availability.
- Downstream Engineering. Like antibodies, sdAb may require affinity maturation, humanization and proper developability. Their simple structure simplifies all these complex procedures, enabling fast track lead optimization. Since they are naturally single chain, they can be incorporated easily in higher structure (bi-specific, tri-specific antibodies). Naturally occurring sdAb are closely related to the human VH3 family, which is the most abundant VH family in humans, making humanization a relatively simple process.
- Small Drug Feature. With regard to targeting, development and administration. sdAb can target the active site of enzymes, receptor clefts and virus hidden epitopes; they can sustain harsher conditions (pH, heat) and be used in formats normally reserved for small drugs (e.g. oral administration).
- Enhanced Tissue Penetration. Better tissue penetration than IgG due to their small size; they can cross natural barriers and are actively under development for crossing the blood brain barrier and targeting the central nervous system. Inversely they also benefit from a better clearance, making them good candidates for tumor imaging.
- Easy Manufacturing. sdAb express very well on a variety of platforms (bacteria, yeast) and are cost-effective to manufacture on a large scale.
Naturally Occurring Single Domain Antibodies
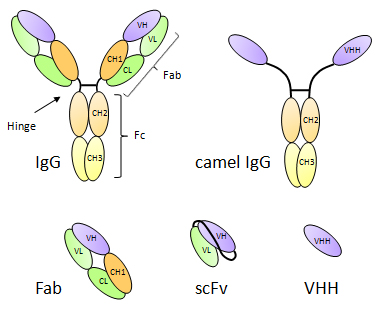
Naturally occurring heavy chain antibodies are found circulating in the serum of camelids (camel, dromedary, llama, alpaca, vicugna). Beside classical full-length antibodies, smaller entities devoid of light chains, still fully functional, were originally discovered in these animals. Due to a defect in an isotype switch in the camelid genome, VHH domains recombine directly with the CH2 domain of the IgG2 and IgG3 isotypes, thus loosing the CH1 domain; the resulting heavy chain antibodies are homodimers of only 75 kD, immunologically relevant because of the presence of the CH2 domain, but having two small VHH domains in place of the two Fab fragments. It is often suggested that the smaller size of VHH domain confers better immunity to viruses by their ability to bind crucial determinants located in deep grooves on the virus surface. Naturally occurring sdAb are homologs with the human VH3 family, the most abundant VH family in human.
Generation of Single Domain Antibodies
L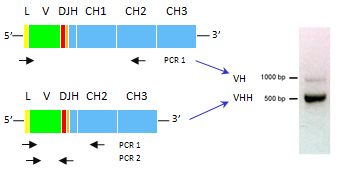 lamas offer an easy and relatively cheap opportunity to generate single domain antibodies. The large animal can undergo multiple antigen stimulation at the same time. Because there is no need to create combinatorial libraries of VH and VL, small phage antibody libraries covering the immunized repertoire can be easily built from circulating B cells and screened rapidly.
lamas offer an easy and relatively cheap opportunity to generate single domain antibodies. The large animal can undergo multiple antigen stimulation at the same time. Because there is no need to create combinatorial libraries of VH and VL, small phage antibody libraries covering the immunized repertoire can be easily built from circulating B cells and screened rapidly.
Because of the high degree of homology between VH and VHH in camelid genome, it is not possible to amplify VHH directly by PCR; instead VHH are rescued through a two-stage PCR where all the heavy chain cDNA are amplified up to the CH2 domain. After isolation of the smaller band containing the VHH, single domain antibodies are re-amplified and cloned in a phagemid vector.
At Antibody Design Labs we routinely achieve >90% VHH after cloning. VHH domains are secondarily confirmed by sequence analysis, showing specific amino acid patterns in frame 2.
Single Domain Antibody Service
 Antibody Design Labs offers a complete line of services for single domain antibody development, from handling animals and immunization at the farm to large scale production of recombinant single domain antibodies.
Antibody Design Labs offers a complete line of services for single domain antibody development, from handling animals and immunization at the farm to large scale production of recombinant single domain antibodies.
Call us immediately to discuss your project directly with our principal scientist.
Service Options
|
Procedure |
Description |
Timeline |
|---|---|---|
|
Immunization |
Standard immunization 12-weeks; short immunization 7-weeks procedure; custom immunization schedule, grouped immunization, dedicated animal available. Blood collection and PBMC preparation at bleed 2 and 3 (50 to 100 ml/bleed). Serum testing by ELISA. |
6-12 weeks |
|
cDNA Preparation |
Isolation of RNA & preparation of cDNA. |
1 week |
|
Library Construction |
Amplification of a large population of VHH. Cloning into any pADL™ phagemid, transformation, preparation of the phage library (>1E8 clones, >95% insert, >80% VHH). QC on 96 clones with full VHH sequence analysis, deep sequencing available. |
4 weeks |
|
Library Screening |
High avidity initial round, secondary selective rounds and competitive rounds using biotin/streptavidin and off-selection, custom selection available. Phage screen in 96-well format by ELISA, multiple ELISA conditions and competitive inhibition available. Phage validation by phage ELISA, DNA sequencing and direct binding assay using free VHH fragment. Deliverable free VHH fragment from best clones. |
4-6 weeks |
|
Production |
Cloning & large-scale expression of VHH. |
custom |
Service Description
Immunization & Animal Handling
Our animal facility is located in South California in San Diego area, which counts among the largest farming facilities for the diagnostic and discovery industry in U.S. Our staff will assist you in determining the best immunization strategy compatible with your time frame. We offer different levels of work to our clients, including access to dedicated “clean” animals. PBMC purifications are processed immediately on site without delay after blood collection to preserve the quality of the mRNA and the high diversity of the library.
Construction of Immunized Single Domain Antibody Library
As one of the leaders in phage display technology, Antibody Design Labs offers all the tools necessary to build the best libraries. Our extended set of primers based on RACE sequencing covers many VHH genes, including sequences unreported in the IMGT database. Construction of libraries follows state-of-the-art procedure with a two-stage amplification of VHH fragments and cloning in any of the pADL™ phagemid vector series. We routinely build libraries with 1E8 clones and above, although the single chain nature of VHH allows for isolation of binders from much smaller libraries, as low as 1E6 members and fewer.
Screening of Single Domain Antibody Library
T he high level of VHH expression usually prevents toxicity issues often seen with the display of Fab fragments and scFv. Good binders can be isolated within a limited number of rounds, although finding clones targeting the proper epitope can be challenging. We can accommodate custom screening tailored to your needs, from cell surface panning to competitive rounds.
he high level of VHH expression usually prevents toxicity issues often seen with the display of Fab fragments and scFv. Good binders can be isolated within a limited number of rounds, although finding clones targeting the proper epitope can be challenging. We can accommodate custom screening tailored to your needs, from cell surface panning to competitive rounds.
Additional Antibody Services
We offer multiple services to streamline the development of the best clones, from large-scale production to cloning and production into mammalian expression system of multi-speficic antibodies. Please, consult with our scientists to determine the best and lowest cost strategy.
Request Service
P
lease, request a specific quote by completing the online quotation form accessible by pressing the following button; you will be contacted back by the next business day.
For all other inquiries, contact us by email at info@abdesignlabs.com or by phone at 1-877-223-3104 (PST). We guarantee competitive pricing.
All our work is performed by highly experienced staff at our San Diego facility in California. We use highly efficient cloning techniques to ensure timely delivery and have very high standards for validation. All products and services are for research use only and are not intended for use in humans.

